ENJAYMO is the first and only approved treatment for Cold Agglutinin Disease (CAD)1
ENJAYMO is a classical complement inhibitor indicated for the treatment of hemolysis in adults with CAD.

ENJAYMO specifically targets and blocks complement-mediated hemolysis in CAD at C1s1
Autoplay
Watch how ENJAYMO works to inhibit complement-mediated hemolysis at C1s1
Cold Agglutinin Disease (CAD): The complement system and ENJAYMO (sutimlimab-jome)
ENJAYMO is a classical complement inhibitor indicated for the treatment of hemolysis in adults with Cold Agglutinin Disease. ENJAYMO is contraindicated in patients with known hypersensitivity to sutimlimab-jome or any of the inactive ingredients.
In this video, we will briefly review the complement system, how CAD affects the complement system, and how ENJAYMO works.
CHAPTER 1: How the complement system works
The complement system, also known as the complement pathway, is part of the innate immune system and functions primarily as a first-line host defense against pathogenic invaders.
The major functions of the complement system include inflammatory cell recruitment, the body’s response to infection or tissue injury; pathogen opsonization, the marking of the surface of pathogens for destruction; pathogen lysis, the breakdown of infectious organisms; and immune clearance, the removal of apoptotic cells from the body.
The complement system is composed of 3 distinct pathways—classical, lectin, and alternative. Each pathway is distinguished by the trigger that activates it.
The classical pathway is initiated typically by antigen-antibody complexes upstream at the C1 complex.
The lectin pathway is initiated when various lectin complexes are activated by binding mannose-binding lectin, or MBL, on pathogens.
The alternative pathway is initiated by factors B and D, which activate upon spontaneous hydrolysis of C3 on pathogens.
The 3 distinct pathways converge with formation of C3 convertase enzyme downstream.
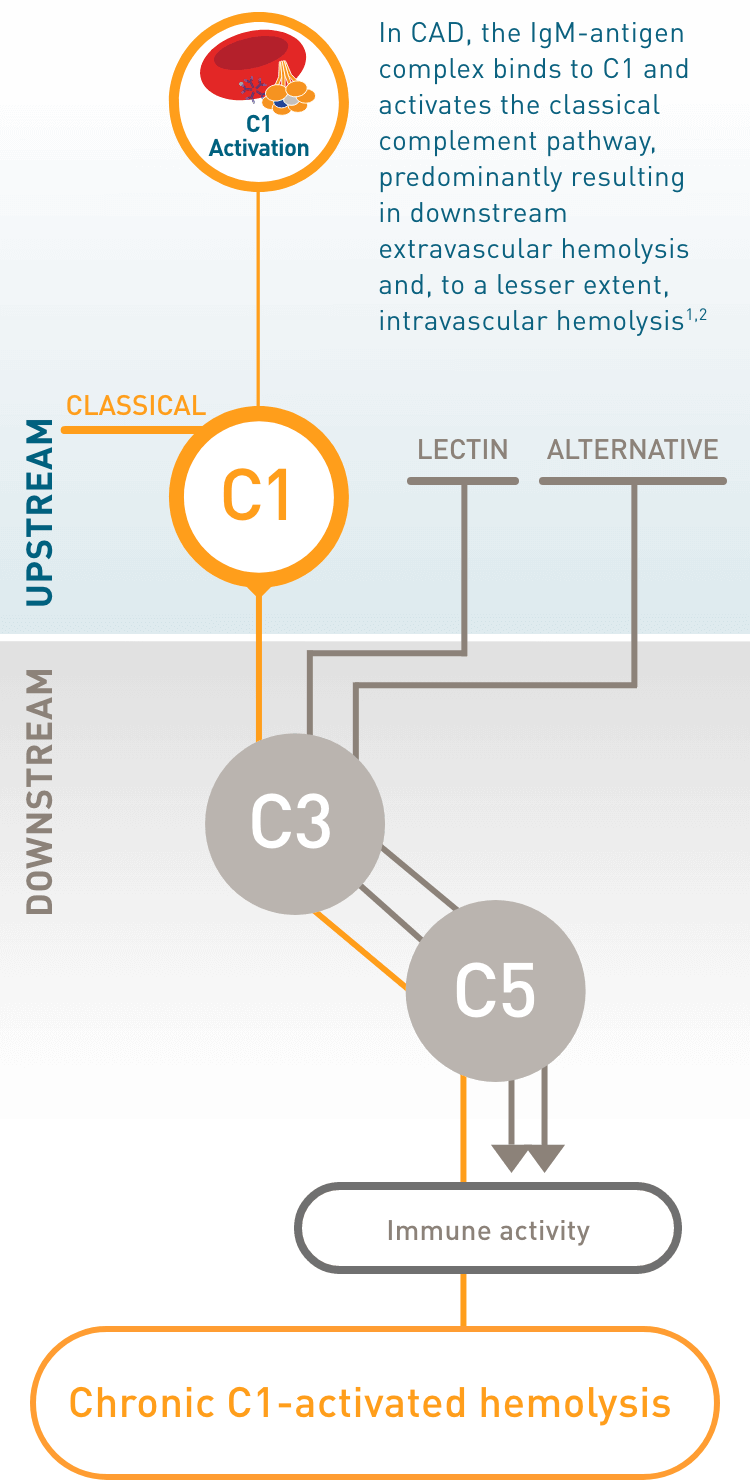
In Cold Agglutinin Disease, activation of the classical complement pathway occurs when autoantibodies, called cold agglutinins, typically IgM, bind to the I antigen on red blood cells.
CHAPTER 2: The classical complement pathway in Cold Agglutinin Disease (CAD)
Cold Agglutinin Disease, or CAD, is a rare, chronic autoimmune hemolytic anemia characterized by C1-activated hemolysis, which is a result of classical pathway activation and persistent complement activation.
In CAD, cold agglutinins, primarily IgM autoantibodies, mistakenly bind to red blood cells, or RBCs, at temperatures less than or equal to 37 °C. IgM autoantibodies are potent classical complement pathway triggers.
The C1 complex—which is made up of subcomponents C1q, C1r, and C1s—is recruited by the cold agglutinin complex and activated upstream by C1s in the classical complement pathway.
Continued C1 activation of the classical complement pathway by cold agglutinins leaves patients in a constant hemolytic state with acute consequences and chronic risk.
The predominant form of chronic hemolysis in CAD is extravascular hemolysis, which occurs in the liver, where C3b-coated cells are phagocytized by macrophages. Intravascular hemolysis occurs to a lesser extent.
Persistent complement activation can also result in the generation of anaphylatoxins C3a and C5a.
CHAPTER 3: The ENJAYMO mechanism of action in Cold Agglutinin Disease (CAD)
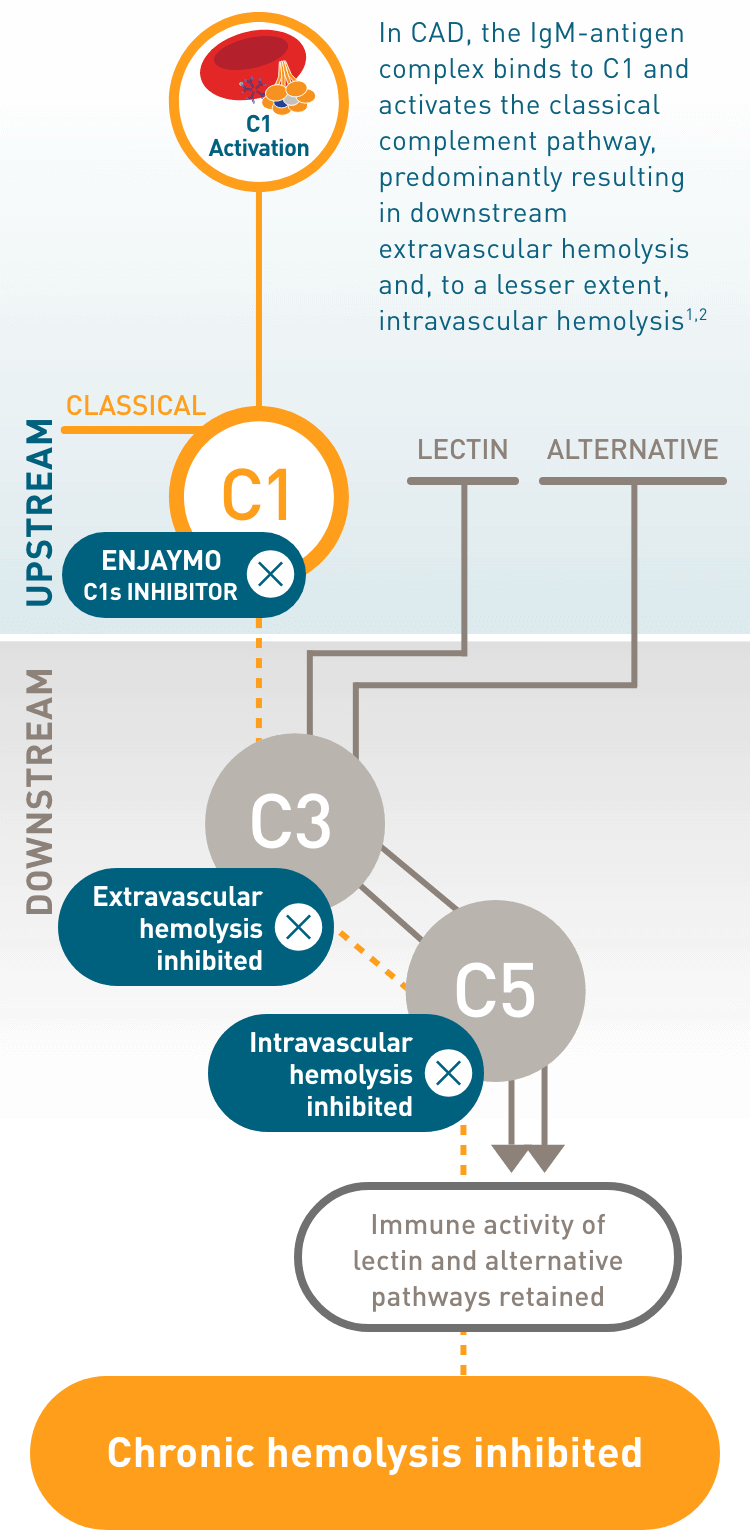
ENJAYMO, the first and only approved treatment for Cold Agglutinin Disease, also known as CAD, inhibits C1-activated hemolysis upstream in the classical complement pathway at C1s.
ENJAYMO® (sutimlimab-jome) is indicated for the treatment of hemolysis in adults with agglutinin disease (CAD).
ENJAYMO, a C1s inhibitor, targets the C1 complex, which is composed of the subcomponents C1q, C1r, and C1s.
Specifically, ENJAYMO inhibits complement protein component 1, s subcomponent, or C1s, in the C1 complex. C1s is a serine protease responsible for activating the classical complement pathway.
By blocking C1s, ENJAYMO prevents downstream activation of the classical complement cascade and deposition of complement opsonins on the surface of RBCs.
Inhibition at C1s prevents the enzymatic action at the C1 complex, thereby addressing the underlying cause of hemolysis in CAD.
The targeted inhibition of C1-activated hemolysis by ENJAYMO, in turn, inhibits extravascular hemolysis, the predominant form of hemolysis in CAD. It also inhibits intravascular hemolysis further downstream.
By specifically targeting C1s upstream in the classical pathway, ENJAYMO blocks C1-activated hemolysis, inhibiting downstream classical pathway activity.
Meanwhile, ENJAYMO preserves the important immune surveillance activities of the lectin and alternative pathways.
ENJAYMO HCP INDICATION AND IMPORTANT SAFETY INFORMATION
INDICATION
ENJAYMO® (sutimlimab-jome) is indicated for the treatment of hemolysis in adults with cold agglutinin disease (CAD).
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
ENJAYMO is contraindicated in patients with known hypersensitivity to sutimlimab-jome or any of the inactive ingredients.
WARNINGS AND PRECAUTIONS
Serious Infections Including Those Caused by Encapsulated Bacteria
- ENJAYMO, a proximal classical complement C1s inhibitor, increases susceptibility to serious infections, including those caused by encapsulated bacteria e.g. Neisseria meningitidis (any serogroup, including non-groupable strains), Streptococcus pneumoniae, and Haemophilus influenzae type B.
- Life-threatening and fatal infections with encapsulated bacteria have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors.
- Serious infections (bacterial and viral) were reported in 15% (10/66) of patients receiving ENJAYMO in the two phase 3 trials. These infections included urinary tract infection with sepsis, respiratory tract infection, pneumonia, otomastoiditis, and skin infections. One patient (1.5%) died due to Klebsiella pneumoniae.
- Complete or update vaccination against encapsulated bacteria at least 2 weeks prior to administration of the first dose of ENJAYMO, according to the most current ACIP recommendations for patients receiving a complement inhibitor.
- If urgent ENJAYMO therapy is indicated in a patient who is not up to date on their vaccine(s), administer as soon as possible.
- Vaccination does not eliminate the risk of serious encapsulated bacterial infections. Closely monitor patients for early signs and symptoms of serious infection and evaluate patients immediately if an infection is suspected.
-
If ENJAYMO treatment is administered to patients with active systemic infections, monitor closely for signs and symptoms of worsening infection. Some infections may become rapidly life-threatening or fatal if not recognized and treated promptly. Inform patients of these signs and symptoms and steps to be taken to seek immediate medical care.
- Consider interruption of ENJAYMO treatment in patients who are undergoing treatment for serious infection.
- Consider patients’ immune status when initiating treatment with ENJAYMO.
Infusion-Related Reactions
- Administration of ENJAYMO may result in infusion-related reactions. In the two phase 3 trials, 29% (19/66) of patients treated with ENJAYMO experienced infusion-related reactions. One patient permanently discontinued ENJAYMO due to an infusion-related reaction.
- Monitor patients for infusion-related reactions and interrupt if a reaction occurs.
- Discontinue ENJAYMO infusion and institute appropriate supportive measures if signs of hypersensitivity reactions, such as cardiovascular instability or respiratory compromise, occur.
Risk of Autoimmune Disease
- Based on its mechanism of action, ENJAYMO may potentially increase the risk for developing autoimmune diseases such as systemic lupus erythematosus (SLE). Development of SLE has been associated with inherited classical complement deficiency.
- In clinical trials, 4.5% (3/66) of patients developed a relapse or worsening of previously diagnosed autoimmune disease.
- Monitor ENJAYMO patients for signs and symptoms and manage medically.
Recurrent Hemolysis After ENJAYMO Discontinuation
- If treatment with ENJAYMO is interrupted, closely monitor patients for signs and symptoms of recurrent hemolysis, eg, elevated levels of total bilirubin or lactate dehydrogenase (LDH) accompanied by a decrease in hemoglobin, or reappearance of symptoms such as fatigue, dyspnea, palpitations, or hemoglobinuria. Consider restarting ENJAYMO if signs and symptoms of hemolysis occur after discontinuation.
ADVERSE REACTIONS
- The most common adverse reactions in the CADENZA trial (Part A) (incidence ≥18%) are rhinitis, headache, hypertension, acrocyanosis, and Raynaud’s phenomenon. The most common adverse reactions in the CARDINAL trial (incidence ≥25%) are urinary tract infection, respiratory tract infection, bacterial infection, dizziness, fatigue, peripheral edema, arthralgia, cough, hypertension, and nausea.
Please see accompanying full Prescribing Information.
Inhibit chronic C1-activated hemolysis upstream in the classical complement pathway by choosing ENJAYMO.
Pharmacodynamic data show inhibiting complement-mediated hemolysis at C1s started with the first dose of ENJAYMO1
In 1 dose, ENJAYMO demonstrated inhibition of complement-mediated hemolysis at C1s
- >1 g/dL increase in hemoglobin†
- Near normalization of bilirubin†
Continued use of ENJAYMO provided complete classical complement pathway inhibition, leading to inhibition of complement-mediated hemolysis evidenced by
- Normalization of bilirubin
- Decrease in LDH
- Increase in haptoglobin
- Decrease in reticulocytes
†Mean Hb at baseline: 9.15 g/dL; mean bilirubin at baseline: 2.41 mg/dL.
ENJAYMO works fast to inhibit complement-mediated hemolysis
90% inhibition of the complement pathway was observed following a single dose of ENJAYMO and sustained when ENJAYMO concentrations were ≥100 mcg/mL1